The US medical devices market has experienced a major change since the introduction of the Food and Drug Administration (FDA) UDI ruling back in September 2014. We spoke with Paul Hammond, Domino’s Medical Device Sector Manager for North America to find out what companies need to do to adapt to this new initiative.
How has the medical device market changed in the US?

When speaking to Paul he confirmed that the biggest change in the US medical device market was the adoption of the FDA UDI initiative. In the past there was very little traceability of medical devices in the US which resulted in issues with patient safety and healthcare systems. Situations occurred where surgeons were operating on patients without having the required product information of the device such as, who made it and where it was manufactured.
The FDA have now released a final ruling requiring that medical devices distributed in the US, carry a unique device identifier, known as a UDI. This data is then submitted by the manufacturers to the FDA Global UDI Database (GUDID).

What is UDI in the US?
Paul explained that the first stage of UDI implementation is focused on the highest risk medical devices.
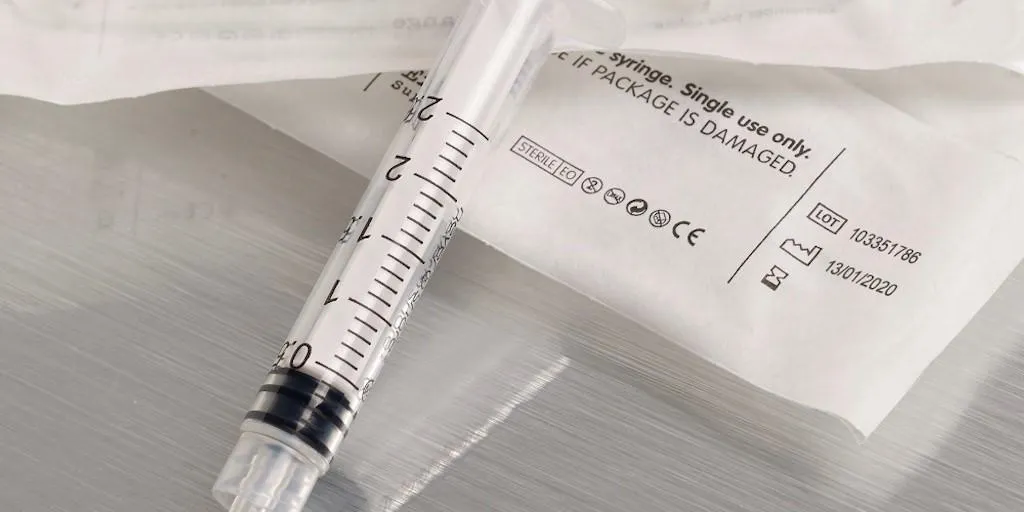
Class III, II, and I are the main classes of devices. Within each class there are different products depending on the importance of the device. For example, the highest risk products are Class III, which include pacemakers and other implantable devices. Class II covers devices such as syringes, catheters and hearing aids. Class I are less severe products, for example, bandages, sutures and devices of that nature.
Companies will need to evaluate their products and their vision systems. They need to review the applicable ISO standards plus review and comply with the GS1 standard. If companies follow the GS1 barcode and labelling standards they should be in a good position to obtain the highest possible barcode grades to meet UDI compliance
Paul Hammond
Even the smallest of medical devices may require a UDI code. Manufacturers will need to apply a tiny 2D Code onto these devices to remain compliant and to show the device is classified as reusable or can be reprocessed. Domino’s laser coding technology can apply a permanent UDI code on devices as small as a spinal screw head.
How has it affected the market?
We asked Paul to explain the affect that this initiative has had on the US medical devices market. In response Paul explained that all manufacturers of medical devices need to review the legislation and plan how they will implement UDI. Larger companies within the US have had teams dedicated to becoming UDI compliant for a while. It is the mid to small-sized manufacturers that are facing the biggest challenges due to a lack of resources, experience and funding. Smaller companies are looking for exemptions because they are struggling to meet the legislation requirements.
Clinics and hospitals in the US will need to have the ability to read and decipher the UDI information on the medical products used and validate against the FDA GUDID database. With this in mind, many hospitals, clinics and healthcare facilities will need to upgrade their inventory and scanning equipment and this takes time to implement.
In Domino’s experience, it typically takes 12 to 18 months for medical device manufacturers to become UDI compliant
Paul Hammond
Domino have the knowledge and expertise to support companies on their journey to becoming UDI compliant. Validation packages are available from Domino which help customers validate their products. This is something companies need to be doing.
Domino’s range of coding and marking solutions enable the printing of UDI barcodes and labels on all packaging levels. The upfront cost of implementing manufacturing lines with new coding and marking solution can be costly. This is why Domino offers its Relax package, which helps companies to implement the necessary equipment, without having to go through a lengthy capital expenditure process.
To find out more about becoming UDI compliant and the upcoming EU Medical Device Regulation, download our white paper today.