March 2023 saw yet another extension of the EU MDR transition period, with the publication of Regulation (EU) 2023/607 in the Official Journal of the European Union. The latest extension gives manufacturers of MDR legacy products longer deadlines to certify their products.
The extension has been granted as a result of capacity restrictions from notified bodies, which are currently working through MDR certifications for products holding certifications issues under Directives 93/42/EEC and 90/385/EEC, and approaching expiration in May 2024.
Bart Vansteenkiste, Global Life Sciences Sector Manager, Domino Printing Sciences, offers medical device manufacturers advice on how to make use of the time remaining.
Recap: What is required from the EU MDR?
The EU MDR replaces the EU’s current Medical Device Directive (93/42/EEC) and Directive on Active Implantable Medical Devices (90/385/EEC).
As part of the new regulations, manufacturers of medical devices for sale within the EU must adhere to strict guidelines to ensure their products are safe to use. The regulations cover all medical devices sold in the EU – everything from scalpels and needles to pacemakers, prosthetic limbs, and medical devices with a cosmetic purpose such as coloured contact lenses.
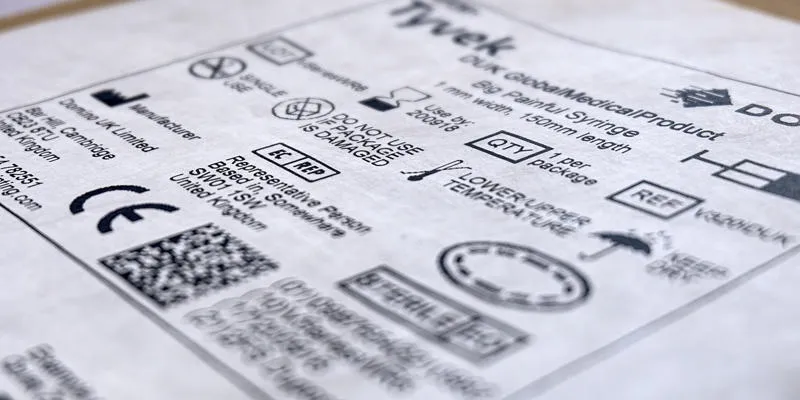
The introduction of the EU MDR obligates medical device manufacturers to invest in technology to enable the fast and accurate application of traceability coding to products and packaging at the individual item level.
Failure to comply with these procedures may mean that devices are withdrawn from sale, with device manufacturers no longer being able to supply their products to other EU member states.
As part of the new regulations, medical devices need to be assigned a unique device identification (UDI) code, and have their UDI recorded, indexed, and registered in EUDAMED, the central European Database for Medical Devices. Publication and full functionality of EUDAMED is expected to be in place by the end of 2024, with mandatory full use for all medical device manufacturers following by 26 May 2026.
Latest announcement
With the latest announcement, and publication of Regulation (EU) 2023/607, the European Commission has extended the EU MDR transition period in an attempt to address the risk of medical device shortages in the EU market.
With some 21,376 products holding certificates issued under Directives 93/42/EEC and 90/385/EEC, and due to expire on 26 May 2024, notified bodies have estimated that they can only issue 7,000 MDR certificates by the deadline, risking disruption to medical device supply.
In response to this Regulation (EU) 2023/607 was introduced, with the following main changes:
- Extension of MDR transition period until 31 December 2027 or 31 December 2028, depending on the risk class of the device
- Introduction of a transition period of until 26 May 2026 for class III custom-made implantable devices
It should be noted that the new EU MDR extension is subject to several conditions to ensure that devices are safe to use.
To benefit from the extension manufacturers must:
- Establish a quality management system compliant with the MDR by 26 May 2024
- Submit a formal application to a notified body for conformity assessment by 26 May 2024
- Arrange conformity assessment with a notified body by 26 September 2024
In addition, the extension only applies to MDR legacy devices that:
- Continue to comply with the relevant directive
- Do not present significant changes in design or intended use
- Do not present unacceptable risks to patients and users
Products with an expired certificate may only benefit from the extension if the manufacturer has concluded a contract with a notified body for conformity assessment; or if the manufacturer has been granted a specific exemption under Article 97 or 59 of the MDR.
Where are we now?
With the EU MDR timeline apt to change, it is important that organisations stay abreast of current developments as and when they arise. Medical device manufacturers should speak to institutions such as GS1, as well as notified bodies and trusted partners for advice and support.
So far, manufacturers have the benefit of a technical bulletin, available from the European Commission, which provides information on how data should be submitted. The technical bulletin is addressed to the different needs of each manufacturer.
As EUDAMED displays a challenging and continuous project for each manufacturer, it is advisable to begin collecting data as soon as possible. Moreover, manufacturers should make themselves familiar with the technical aspects of gathering and uploading their data to EUDAMED. Manufacturers will need to collect data on each product according to the Annex VI, Part B of the EU MDR, and begin preparing the data for sharing on EUDAMED.
As well as preparing all data in advance of the revised deadline, manufacturers should ensure that they have the right partners who support them through the process. To ensure that their devices comply with the new regulations, manufacturers should speak to their code issuing agency and notified bodies for advice.
At present, some large medical device manufacturers are utilising up to 25% of their employee base for bringing their procedures up to standard. Small- and medium-sized manufacturers are unlikely to have the capacity to dedicate so much of their workforce and should consider options for external support.
Issuing agencies
In June 2019, GS1 became the first issuing agency for EU MDR compliant codes, meaning that 2D DataMatrix and GS1-128 codes can be used going forward. Further issuing agencies followed suit after, with potential for the use of HIBCC, ICCBBA, and IFA coding.
Notified bodies
As part of existing EU Directives (90/385/EWG) and (93/42/EWG) manufacturers audit and check their products on a regular basis to ensure compliance. Notified bodies support manufacturers in this process to ensure that new and existing products can be sold. Of the 57 notified bodies across Europe dealing with current legislation, only 37 have applied for accreditation to the EU MDR.
At the time of writing, the landmark figure of 30 notified bodies designated under the MDR had been reached, however, that the latest extension is due to capacity restrictions of the notified bodies suggests that more are badly needed to bring the regulation to completion.
Manufacturers can decide which notified body they choose for their products but should bear in mind that some existing notified bodies will no longer be available for assistance under the EU MDR. The new process of designation is significantly more challenging, and as such, around 20 notified bodies have not applied.
A global effect
The EU MDR covers all items sold within the EU, but this does not mean that only EU member states need to fulfil the requirements. All manufacturers that wish to sell their product in the EU need to ensure that they satisfy the EU MDR requirements, or they may see their products removed from sale.
Equally, manufacturers who are registered according to the EU MDR should focus future attentions on ensuring compliance in different parts of the world where additional legislation may be a factor. At present, the US have their legislation. China, Switzerland, Egypt, Australia, Saudi Arabia and Brazil are in the process of passing legislation on the identification of medical devices, with further discussions currently in progress in Colombia, Japan, Singapore, and UK.
Who can offer further advice and support?
With the deadline for ensuring compliance with the EU MDR fast approaching, the time to act is now.
The experts at Domino are on hand to help you ensure that your products are compliant with the upcoming EU MDR. At Domino, we provide validation packages to help manufacturers authenticate their products and prepare for EU MDR accreditation.
Our range of coding and marking solutions offers manufacturers of medical devices the most suitable technology in order to achieve UDI compliance. For advice and support in how to ensure your coding is EU MDR compliant, contact the experts at Domino.